|
(iii) Elements revolve around the nucleus in certain definite circular paths called orbits or shells. (ii) The third-period elements, Na, Mg, Al, Si, P and Cl summarize the properties of their respective groups and are called typical elements. (i) The properties that reappear at regular intervals, or in which there is a gradual variation at regular intervals, are called periodic properties and the phenomenon is known as the periodicity of elements. (iv) Noble gases are placed at the extreme right of the periodic table. (iii) Copper and zinc are transition elements. (ii) Valency depends upon the number of valence electrons in an atom. (i) Elements in the same group have equal valency. (iv) Transition elements are placed at the extreme right of the periodic table. (iii) Copper and zinc are representative elements. 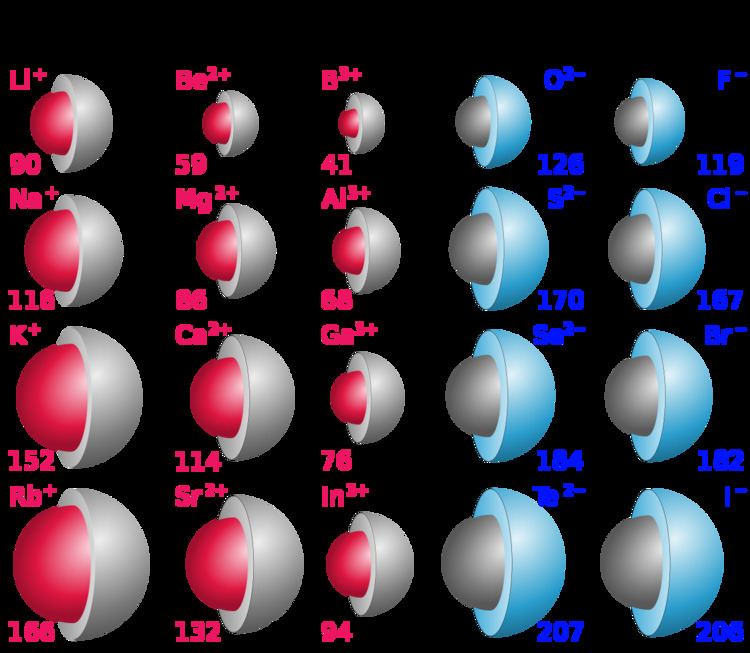
(ii) Valency depends upon the number of shells in an atom. (i) Elements in the same period have equal valency. But the valency increases only up to Group 14, where it becomes 4, and then it decreases, that is, it becomes 1 in Group 17. In a given period, the number of electrons in the valence (outermost) shell increases from left to right. If the number of electrons present in the outermost shell is 7, then its valency is again 1 (8 – 7 = 1) as it can accept 1 electron from the combining atom. If the number of electrons present in the outermost shell is 1, then it can donate one electron while combining with other elements to obtain a stable electronic configuration. Valency depends on the number of electrons in the outermost shell (i.e. Group 1 elements have 1 electron in their outermost orbital, while Group 7 elements have 7 electrons in their outermost orbital. It is just a number and does not have a positive or negative sign. It is equal to the number of electrons an atom can donate or accept or share. Valency is the combining capacity of the atom of an element. Question 5Įlements of group 1 and elements of group 17 both have valency 1. one by one but the number of valence electrons remains the same. (ii) While going from top to bottom in a group, the number of shells increases successively i.e. (i) Though the number of shells remain the same, number of valence electrons increases by one, as we move across any given period from left to right. How does electronic configuration in atoms change Periodicity is observed due to the similar electronic configuration. (number of valence electrons/ atomic number/ electronic configuration) Answer 3 Periodicity is observed due to the similar…………. 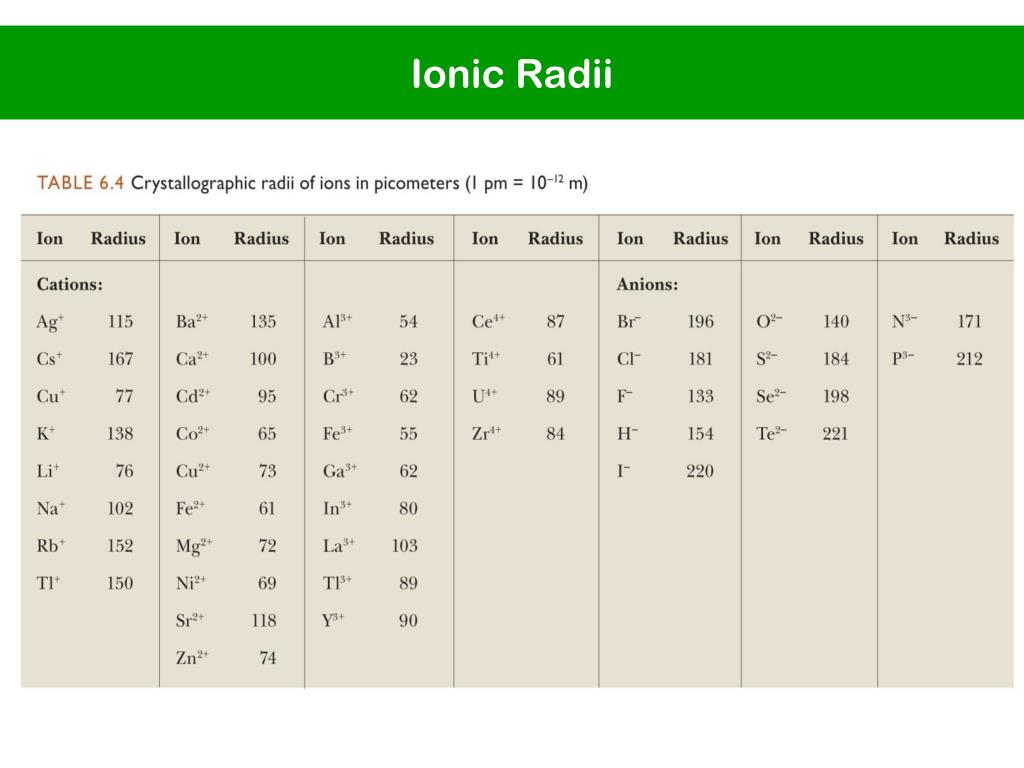
The horizontal rows are known as periods and vertical columns in the periodic table are known as groups. What are horizontal rows and vertical columns in a periodic table known as? Answer 2 (iii) Modern Periodic table has 7 periods and 18 groups. 
(ii) Henry Moseley put forward the modern periodic law. (i) The modern periodic law states that “The properties of elements are the periodic functions of their atomic number.” (iii) How many groups and periods does modern periodic table have? Answer 1 (ii) Name the scientist who stated the law. TM ions provide the glasses stable behavior to irradiation because of their flexible configuration to heal radiation defects affording the possible use of glasses as radiation shielding materials (up to 5 kGy).Intext-1, Periodic Table Selina Solution Chapter 1 for ICSE Concise Selina Chemistry Class 10 AC conductivity, dielectric constants and dielectric loss revealed rising with incorporating TM ions and irradiation. Trigonal BO 3 units appeared more extensive than tetrahedral BO 4 on FTIR spectra after irradiation. Chemical durability in H 2O, HCl, and NaOH revealed good behavior from 15 to 35 days. ESR spectra revealed values of g// ∼ 2.01 and g┴ ∼ 2.025 correlated to the transition metal (TM) ion valence state. Optical energy gap (E opt) appeared dependent on glass density in a direct relation. UV–visible spectra revealed Co 2+ (3d 7) in octahedral and tetrahedral coordinations, while Ni 2+ (3d 8) and Fe 2+ (3d 6) favored the octahedral coordination. Co 2+/ Ni 2+/ Fe 3+ borate glasses were prepared by melting-annealing procedure and extensively characterized towards gamma irradiation (up to 8 kGy).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
